The word arose when a proofreader's question about the symbol for density, "D or d?" was taken literally. Webster's Second International Dictionary, 1934 showed Dord as a synonym for density. It's a ghost word, an accidental creation that came to be reproduced in dictionaries. It moves to the numerator, giving us the volume unit we desire for the answer. Note that, in the g/cm 3 unit, that the cm 3 is in the denominator of the denominator. The water level rises to the 49.1 mL mark, From this information, calculate the density of iron.ġ) Calculate the volume of the iron shot:Įxample #10: What volume of silver metal will weigh exactly 2500.0 g. (4.50 cm) (5.20 cm) (6.00 cm) = 140.4 cm 3 (ignore sig figs for the moment)ġ591 g / 140.4 cm 3 = 11.3 g/cm 3 (3 sig figs because of the length measurements on the lead block)Įxample #9: 28.5 g of iron shot is added to a graduated cylinder containing 45.5 mL of water. From this information, calculate the density of lead.ġ) Determine the volume of the lead block: The density of benzene is 0.8786 g/mL.Įxample #8: A block of lead has dimensions of 4.50 cm by 5.20 cm by 6.00 cm. From this information, calculate the density of carbon tetrachloride.Įxample #6: Calculate the density of sulfuric acid if 35.4 mL of the acid weighs 65.14 g.Įxample #7: Find the mass of 250.0 mL of benzene. The weight of the flask and carbon tetrachloride is found to be 703.55 g. Significant figures in the answer are dictated by the length measurements of two sig figs.Įxample #5: A flask that weighs 345.8 g is filled with 225 mL of carbon tetrachloride. From this data, what is the density of copper? The dimensions of the block are 8.4 cm by 5.5 cm by 4.6 cm. From this information, calculate the density of mercury.Įxample #3: What is the mass of the ethyl alcohol that exactly fills a 200.0 mL container? The density of ethyl alcohol is 0.789 g/mL.Įxample #4: A rectangular block of copper metal weighs 1896 g. The mercury used to fill the cylinder weighs 306.0 g. What is its density?Įxample #2: Mercury metal is poured into a graduated cylinder that holds exactly 22.5 mL. You can also check it out by way of the units: (g / cm 3) x cm 3 cancels out the volume unit leaving grams, the desired unit for mass.Įxample #1: A block of aluminum occupies a volume of 15.0 mL and weighs 40.5 g. This leave you with dV (ignore the fact that it is in the denominator). Simply cover up whichever value you need to calculate and the other two are shown in their proper placement, be it to multiply or to divide.įor example, cover up the M. This reminder might help you in figuring out how to solve density problems: This means that sometimes the density of a liquid is described using cm 3 and the density of a solid is described using mL. Since one mL equals one cm 3, there is no functional difference between g/cm 3 and g/mL. Solids: d = grams ÷ cubic centimeters (cm 3 is the symbol for cubic centimeters) The unit for cubic centimeters is cm 3 and for milliliters is mL. Note the difference in units in the formulas of the density of a solid and liquid. 
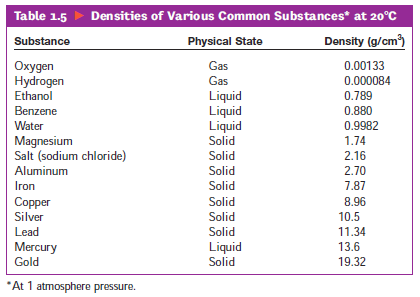
We will ignore these small amounts and act as if all our density problems are at the same temperature and pressure. Although the density of liquids and solids do change with temperature and pressure changes, the amount is fairly small. The density of a gas will be dealt with in a later unit, because its density is very sensitive to temperature and pressure. We can calculate the density of a solid, liquid, or gas. However, a lower-case d is more often used in intoductory settings like the one you are currently reading. Oftentimes, the rho shape that a textbook would use looks more like the lower-case letter p. This is shown in equation form, as follows:īy the way, the lower-case Greek letter rho, ρ, is also used to symbolize density. Density is calculated by dividing the mass of an object by its volume. Density (whose most common symbol is the lowercase letter d) is defined as mass per unit volume. There are physical characteristics of a substance that help identify the substance. Twenty Examples Probs #1-10 Probs #11-25 Probs #26-50 All the examples & problems, no solutions Significant Figures Menu
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
